X-ray Transmission
X-ray Transmission / Absorption Gauging
Basic XRT / XRA Theory for Sulfur Analysis in a Petroleum Process Stream
X-ray Transmission (XRT) gauging has long been an accepted technique for the measurement of sulfur (S) in heavy hydrocarbon process streams. Whether used for pipeline switching, crude oil blending or to assay or blend marine and bunker fuels, the Rigaku NEX XT XRT process analyzer is well suited to rigorous process environments, with pressures up to 1480 psig and temperature up to 200°C.
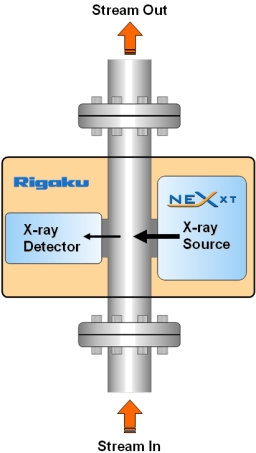
X-ray transmission gauging involves measuring the attenuation of a monochromatic X-ray beam at a specific energy (21 keV) that is specific to sulfur (S). In practice, a process stream passes through a flowcell where sulfur (S), in the hydrocarbon matrix, absorbs X-rays transmitted between an X-ray source and detector (see schematic at left). The recorded X-ray intensity is inversely proportional to the sulfur concentration, thus the highest sulfur levels transmit the least X-rays.
Transmission of X-rays through the flowcell is given by the following equation:
T=I/Io=exp-dt[μm(1-Cs)+ μsCs]
where:
I = measured X-ray intensity (after flowcell, in photons/sec)
Io = initial X-ray intensity (before flowcell, in photons/sec)
d = density of the hydrocarbon stream
t = thickness of the flowcell path (in cm)
μm = molar absorptivity for the hydrocarbon matrix @ 21 keV (cm2/gm)
μs = molar absorptivity for sulfur @ 21 keV (cm2/gm)
Cs = weight fraction of sulfur (% wt/wt)
| Element | Molar Absorptivity @ 21 keV |
| H | 0.37 |
| C | 0.41 |
| O | 0.79 |
| S | 5.82 |
As illustrated in the table, 21 keV is chosen as the energy of X-rays employed in the measurement because: 1) the molar absorptivities of C and H are almost identical and 2) the absorption due to sulfur is 14X larger than the CxHy matrix and 7X larger than oxygen. Thus the method is insensitive to changes in the C:H ratio and is primarily sensitive to only the sulfur content.
